Co-Diagnostics, Inc. Announces Recent Grant Awards

The Company intends to use funds awarded by the Bill & Melinda Gates Foundation toward completion of tuberculosis and HPV tests for its Co-Dx PCR Home™ platform
SALT LAKE CITY, July 18, 2023 /PRNewswire/ — Co-Diagnostics, Inc. (Nasdaq: CODX)[1] (the “Company” or “Co-Dx”), a molecular diagnostics company with a unique, patented platform for the development of molecular diagnostic tests, today announced grants awarded by the Bill & Melinda Gates Foundation related to the tuberculosis (TB) and human papillomavirus (HPV) tests on the Company’s Co-Dx PCR Home™ platform.
[1] https://www.prnewswire.com/news-releases/co-diagnostics-inc-announces-recent-grant-awards-301879355.html#financial-modalThe TB and HPV grants in the amounts of $1.33 million and $987,000 respectively will be applied towards completion of these tests, intended for priority global markets.

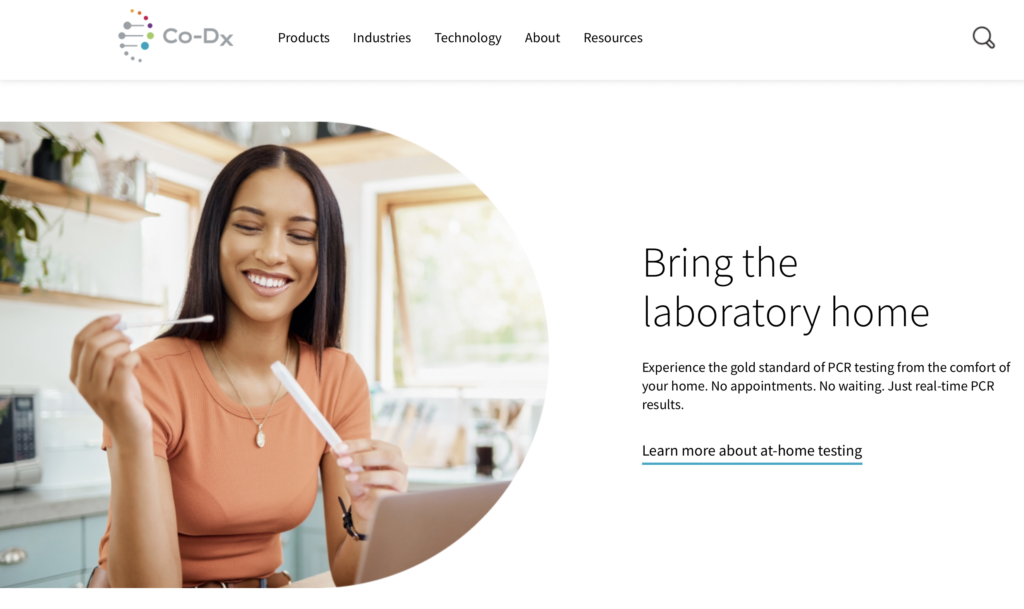
Co-Dx intends to use funds awarded by the Gates foundation toward completion of TB and HPV tests for its new platform
In 2014, all United Nations Member States and the World Health Organization (WHO) committed to ending the global TB epidemic by 2030, before the COVID-19 pandemic slowed, stalled or reversed progress of global TB targets. The WHO estimates that roughly 4.2 million cases of TB went unidentified in 2021 worldwide, representing nearly 40% of actual cases, highlighting the need for a dramatic increase of affordable, high-quality, point-of-care TB diagnostics to enable rapid treatment decisions to be made for a disease that has a mortality rate of about 50% if left untreated.
The National Cancer Institute estimates that high-risk HPVs cause roughly 5% of all cancers worldwide, with the rate among women being nearly 10 times higher than that for men and leading to an estimated 264,000 cervical cancer deaths each year according to the WHO. More than 85% of these deaths are in low- and middle-income countries, and all of which the WHO believes can be dramatically reduced by access to diagnostics, vaccinations, and cancer screenings.
“The WHO and UN initiative to end the global TB epidemic is exactly in line with the key functionality of our new Co-Dx PCR Home platform currently in development, and with our stated mission to prevent the spread of infectious diseases by increasing the availability of high-quality PCR diagnostics worldwide,” said Dwight Egan, CEO of Co-Diagnostics.
“Despite being a highly treatable disease, TB took the lives of 1.6 million people in 2021, and we believe our new platform will help support the worldwide effort to help prevent the spread of both TB and HPV in countries where they have been endemic killers for generations. This includes India, where we have already created infrastructure and manufacturing capabilities through our joint venture, CoSara Diagnostics [2]. We believe that our new platform has the potential to dramatically improve access to diagnostics and thus reduce healthcare costs by making high quality diagnostics more accessible to the people and places where they are most needed.”
[2] https://c212.net/c/link/?t=0&l=en&o=3921847-1&h=3780937680&u=https%3A%2F%2Fcodiagnostics.com%2Fabout%2Fcosara-diagnostics-private-limited%2F&a=CoSara+DiagnosticsThe Co-Dx PCR Home and its associated tests are subject to review by the FDA and/or other regulatory bodies and are not yet available for sale.
Six Technologies and Innovation (Women’s Health Technology) – Bill & Melinda Gates Foundation





Ref: https://www.gatesfoundation.org/ideas/science-innovation-technology/future-womens-health-technology
About Co-Diagnostics, Inc.:
Co-Diagnostics, Inc., a Utah corporation, is a molecular diagnostics company that develops, manufactures and markets state-of-the-art diagnostics technologies. The Company’s technologies are utilized for tests that are designed using the detection and/or analysis of nucleic acid molecules (DNA or RNA). The Company also uses its proprietary technology to design specific tests for its Co-Dx PCR Home™ platform and to locate genetic markers for use in applications other than infectious disease.
Forward-Looking Statements:
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 (PSLRA) that are subject to a number of risks and uncertainties. Forward-looking statements can be identified by words such as “believes,” “expects,” “estimates,” “intends,” “may,” “plans,” “will” and similar expressions, or the negative of these words. Such forward-looking statements are based on facts and conditions as they exist at the time such statements are made and predictions as to future facts and conditions. Forward-looking statements in this release include statements regarding our belief that our new platform will help support the worldwide effort to help prevent the spread of both TB and HPV and our believe that our new platform has the potential to dramatically improve access to diagnostics and reduce healthcare costs. Actual results may differ materially from those contemplated or anticipated by such forward-looking statements. Readers of this press release are cautioned not to place undue reliance on any forward-looking statements. There can be no assurance that any of the anticipated results will occur on a timely basis or at all due to certain risks and uncertainties, a discussion of which can be found in our Risk Factors disclosure in our Annual Report on Form 10-K, filed with the Securities and Exchange Commission (SEC) on March 16, 2023, and in our other filings with the SEC. The Company does not undertake any obligation to update any forward-looking statement relating to matters discussed in this press release, except as may be required by applicable securities laws.
SOURCE: Co-Diagnostics, PRNewswire, Co-Dx-Image
Also Read:
