Highlights:
- On May 4, 2025, India launched two gene-edited rice varieties, DRR Rice 100 (Kamala) and Pusa DST Rice 1.
- The Coalition for GM Free India opposed the introduction, citing potential harm to human health and the environment.
- Critics argue gene editing poses similar risks as GM crops and demand strict regulations.
- Prominent scientists, including Dr. Pushpa Bhargava, warn against GM crops and gene editing, emphasizing the need for public interest protection.
- Vandana Shiva, a well-known environmental activist and scholar, has raised concerns about the potential risks linked to these crops.
- The European Court ruled that gene-edited crops should be regulated like GMOs, highlighting safety concerns.
So, there was this meeting recently between Shivraj Singh Chouhan and Bill Gates, and they really put a spotlight on India’s push for innovative agricultural tech—like genome-edited rice. Pretty fascinating, right? I mean, it shows that India is ready to dive into some of the latest advancements in farming. But, here’s the thing: it also brings up some big questions about safety and ethics. Like, what does it really mean to start introducing genetically modified organisms (GMOs) into our food supply?

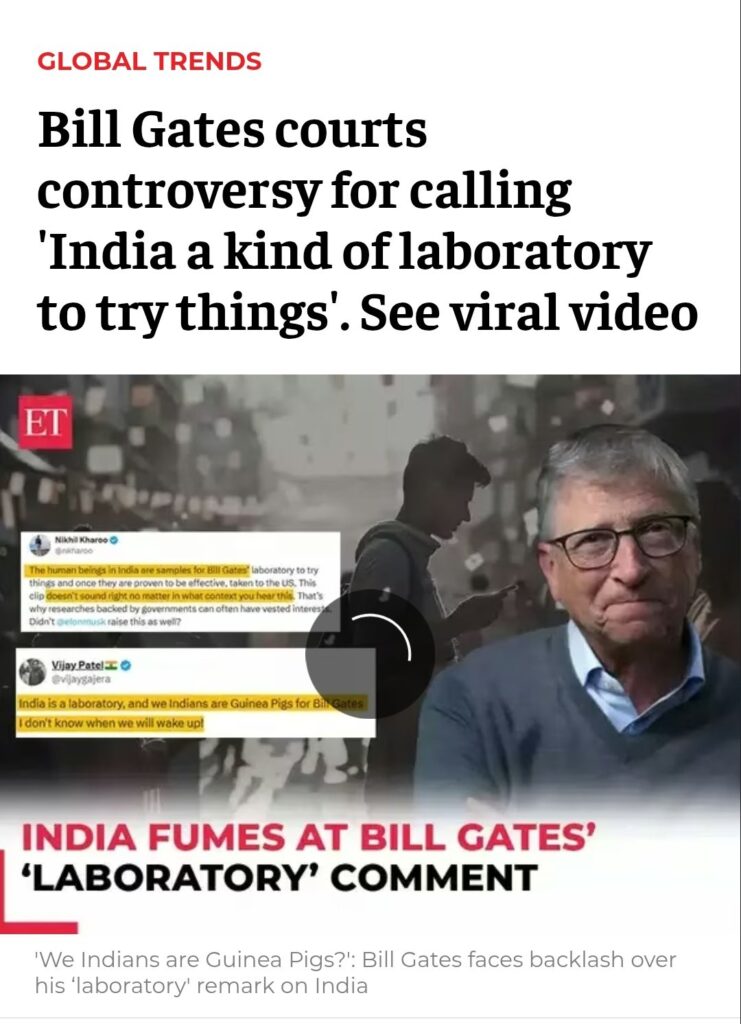
India has historically maintained a firm stance against GM crops, successfully resisting corporate pressure to introduce them into food crops. Instead, GM crops have been limited to cotton production in the country. However, the government’s recent decision to release gene-edited rice varieties has sparked controversy and opposition from groups such as the Coalition for GM Free India.
The Coalition for GM Free India argues that these new crops could potentially pose risks to human health and the environment. They advocate for stringent regulation and emphasize the need to protect indigenous rice varieties, which hold cultural and agronomic significance in the country.
Dr. Pushpa Bhargava, a well-known scientist from India, has made it clear that there is a lot of evidence showing that GM crops are not good. He started the Centre for Cellular and Molecular Biology and was also the Vice Chairperson of the National Knowledge Commission. The Supreme Court of India chose him to observe the Genetic Engineering Appraisal Committee because he is seen as a top expert with a strong commitment to public interest. In an article for The Hindustan Times, he mentioned that over 500 trustworthy research papers show that GM crops can harm human, animal, and plant health, as well as the environment. For instance, a recent study by Indian scientists found that the Bt gene in cotton and brinjal can stop plants from growing properly. In contrast, most studies that support GM crops come from scientists who might have conflicts of interest or whose honesty is questionable.
In an article called ‘Food Without Choice’ in The Tribune, Professor Pushpa Bhargava highlighted how a small but powerful group is pushing for genetically modified crops to benefit themselves and big companies, especially from the US. He warned that this effort, led by Monsanto in India, aims to take control of Indian farming and food production. Since 60% of our population works in agriculture and lives in villages, this would threaten not just our food security but also the safety of farmers and rural communities. Many other respected scientists around the world agree with Dr. Bhargava. A group known as the Independent Science Panel has clearly stated that GM crops have not delivered the benefits they promised and are causing more problems on farms. They also pointed out that contamination from GM crops is unavoidable, meaning GM and non-GM farming cannot coexist. Most importantly, GM crops have not been proven safe, and there is enough evidence to raise serious health and environmental concerns. Therefore, GM crops should be rejected now.
Vandana Shiva, a prominent environmental activist and scholar, has been vocal about the potential dangers associated with these crops.
Claims of Safety vs. Evidence of Harm
Proponents of genetically modified organisms (GMOs) argue that they are necessary for increasing agricultural productivity and ensuring food security. However, Shiva challenges this narrative by highlighting various studies that suggest GMOs may pose significant health risks. For instance, she points out that the introduction of Bt cotton in India was accompanied by a rise in farmer suicides linked to crop failures and debt incurred from purchasing expensive seeds and pesticides. This correlation raises concerns about the broader implications of relying on genetically engineered crops for food production.
Shiva argues that GMOs often lead to increased pesticide use rather than reducing it. For example, while Bt cotton is designed to be pest-resistant, its cultivation has led to the emergence of “superweeds” that require even more chemical interventions. This cycle not only threatens farmers’ livelihoods but also poses risks to consumer health due to increased exposure to harmful chemicals.
Specific Health Concerns
Shiva emphasizes several specific health concerns related to GE crops:
Environmental Impact: The environmental degradation caused by industrial agriculture affects human health indirectly through pollution and loss of biodiversity. The use of herbicides like glyphosate has been linked to various health issues, including cancer. The International Agency for Research on Cancer (IARC) classified glyphosate as probably carcinogenic in humans based on sufficient evidence from animal studies.
Toxicity: There is evidence suggesting that genetically modified foods can lead to adverse health effects in humans. A notable study indicated that rats fed a diet containing GM corn developed tumors and suffered organ damage compared to those fed non-GM diets.
Nutritional Deficiencies: The focus on high-yielding varieties often comes at the expense of nutritional quality. Shiva argues that traditional farming practices yield diverse crops rich in nutrients, whereas monocultures promoted by GE agriculture can lead to deficiencies in essential vitamins and minerals.
The European Court’s ruling that gene-edited crops should be regulated like GMOs further underscores the need for robust safety measures to protect consumers and the environment.
In July 2018, the European Court of Justice (ECJ) delivered a significant ruling regarding gene-edited crops, determining that they should be regulated under the same stringent laws that govern genetically modified organisms (GMOs). The court’s decision stemmed from concerns about the potential risks associated with gene editing techniques, such as CRISPR, which can alter the genetic makeup of organisms in ways that may pose threats to human health and the environment.
The ruling emphasized that gene editing, despite being perceived as a more precise and potentially safer method than traditional genetic modification, still carries inherent risks similar to those associated with GM crops. As a result, the ECJ mandated that any organism produced through gene editing must undergo the same rigorous safety assessments and regulatory processes as GMOs, ensuring that public health and environmental safety are prioritized.
This landmark decision has significant implications for the agricultural biotechnology sector in Europe, as it reinforces the precautionary principle in the regulation of new biotechnological innovations. The ruling aims to maintain transparency and public trust in food safety, while also addressing the ethical and environmental concerns surrounding genetic engineering in agriculture.
Ref: https://enveurope.springeropen.com/articles/10.1186/s12302-018-0182-9
Bill Gates has invested in Editas Medicine, a company focused on gene editing using CRISPR technology. Editas Medicine is using CRISPR-Cas9 to develop new treatments for diseases by directly modifying human DNA.
Editas Medicine and CRISPR Technology
Editas Medicine is a biotechnology company that specializes in gene editing, particularly through the use of CRISPR technology. Founded in 2013, it has been at the forefront of developing therapies aimed at treating genetic diseases by directly modifying human DNA. The company’s approach primarily utilizes the CRISPR-Cas9 system, which allows for precise alterations to the genome.
CRISPR-Cas9 Technology
CRISPR-Cas9 is a revolutionary gene-editing tool that enables scientists to make precise changes to DNA within living organisms. This technology was adapted from a natural defense mechanism found in bacteria, which use it to fend off viral infections. The CRISPR system consists of two key components:
- Guide RNA (gRNA): This RNA molecule is designed to match a specific DNA sequence in the target gene.
- Cas9 Protein: This enzyme acts as molecular scissors that cut the DNA at the location specified by the gRNA.
Drawbacks of CRISPR Technology in Gene Editing
1. Off-Target Effects
One of the primary concerns with CRISPR-Cas9 is the possibility of off-target effects. This occurs when the CRISPR system inadvertently modifies genes other than the intended target. Such unintended edits can lead to harmful consequences, including the activation of oncogenes (genes that can lead to cancer) or disruption of essential genes, potentially causing new health issues rather than resolving existing ones.
2. Ethical Considerations
The use of gene editing technologies raises profound ethical questions. The ability to alter human DNA brings up concerns about “designer babies,” where genetic modifications could be made for non-therapeutic enhancements such as intelligence or physical appearance. This could exacerbate social inequalities and lead to a new form of eugenics.
3. Delivery Mechanisms
Effective delivery of CRISPR components into target cells is another hurdle. Current methods often rely on viral vectors or lipid nanoparticles, which can pose risks such as immune responses or inadequate delivery efficiency. Ensuring that CRISPR components reach the right cells in sufficient quantities without triggering adverse reactions remains a critical area of research.
4. Long-Term Effects and Safety
The long-term effects of gene editing are not fully understood. Since CRISPR alters the genome, any changes made could have unforeseen consequences over time, including potential impacts on future generations if germline cells (sperm and eggs) are edited. There is ongoing debate about whether it is safe to edit human embryos or germline cells due to these uncertainties.
5. Technical Limitations
While CRISPR-Cas9 is powerful, it is not infallible. The technology has limitations regarding the types of mutations it can effectively correct; for example, large deletions or complex genomic rearrangements may not be easily addressed using current CRISPR techniques.
Human Health Effects of Genetically Engineered Crops
Genetically engineered (GE) crops, also known as genetically modified organisms (GMOs), have been a topic of significant debate regarding their safety and potential health effects on humans. The introduction of foreign genes into the genomes of crops government says that it aims to enhance traits such as pest resistance, herbicide tolerance, and nutritional content. However, concerns persist about the implications these modifications may have on human health.
Potential Health Risks
- Allergic Reactions: One of the primary concerns with GE foods is the potential for new allergens to be introduced into the food supply. Genetic engineering can inadvertently transfer allergenic proteins from one food to another. For example, a study published in the New England Journal of Medicine found that soybeans engineered with a Brazil nut gene caused allergic reactions in individuals sensitive to nuts. This risk underscores the need for rigorous testing before GE foods are approved for consumption.
- Toxicity: There are concerns that genetic modifications could lead to unintended toxic effects in food products. The U.S. Food and Drug Administration (FDA) has acknowledged that genetic engineering can create instability in food products, potentially leading to increased levels of naturally occurring toxins or even new toxic compounds. Some studies have indicated that certain genetically modified crops might produce harmful substances that were not present in their non-modified counterparts.
- Antibiotic Resistance: Many GE crops contain antibiotic resistance marker genes used during the development process. There is concern that these genes could transfer to pathogenic bacteria in humans, potentially rendering some antibiotics ineffective. This issue raises significant public health concerns, particularly as antibiotic resistance becomes an increasingly critical global health challenge.
- Cancer Risk: Some research has suggested a possible link between GE foods and cancer due to changes in hormone levels or other biological markers associated with cancer development. For instance, milk from cows treated with recombinant bovine growth hormone (rBGH) has been shown to contain elevated levels of insulin-like growth factor-1 (IGF-1), which is associated with increased cancer risk.
- Nutritional Changes: Genetic modification can alter the nutritional profile of foods, sometimes reducing their nutritional value rather than enhancing it. Studies have indicated that certain GE crops may exhibit undesirable alterations in nutrient levels, which could impact overall dietary quality.
Ref:
- New England Journal of Medicine study on allergenic reactions from GM soybeans [ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7061863/ ]
- FDA’s acknowledgment of genetic instability issues [ https://www.centerforfoodsafety.org/issues/311/ge-foods/ge-food-and-your-health ]
- Concerns over antibiotic resistance from GMOs [ https://www.centerforfoodsafety.org/issues/311/ge-foods/ge-food-and-your-health]
- Research linking rBGH milk consumption with cancer risk [ https://www.centerforfoodsafety.org/issues/311/ge-foods/ge-food-and-your-health]
- Studies indicating nutritional changes in GM foods [ https://www.medicalnewstoday.com/articles/324576 ]
- Ongoing research needs regarding long-term effects [ https://enveurope.springeropen.com/articles/10.1186/s12302-021-00578-9 ]
Source: YouTube, Twitter,
Also Read:
