“COVID-19, AIDS, SARS, MERS, ZIKA and Bird Flu…they all have something in common: the hand of Dr. Anthony Fauci. Every pandemic is exploited to propagate misinformation about vaccines and other treatments – this must not be tolerated any longer.”
Related Article:

What is MERS? According to World Health Organisation (WHO) MERS-CoV (Middle Eastern Respiratory Syndrome Coronavirus).
An overview of how mRNA technology has developed in communist China
Summary: Communist China has been planning mRNA technology development since long ago. The regime acquired mRNA technology in 2016 and engaged in MERS mRNA vaccine development in 2017.
On March 22, 2023, the mRNA Covid vaccine by CSPC Pharmaceutical Group was granted Emergency Use Authorization in Communist China, eleven months after the start of clinical trials. [1] This news aroused great interests worldwide. What is Communist China’s research and development capacity in mRNA technology? This article will provide an overview of Communist China’s mRNA technology development.
Early trailblazers in the mRNA field included Fudan University, the Academy of Military Medical Sciences (AMMS), CanSino Bio, and about nine startups—Abogen Biosciences, LiveRNA (acquired by AIM Bio), RNACure Biopharma, InnoRNA, ImmoRNA Biotechnology, Rhegen Biomedical Technology, Longuide, Stemirna Therapeutics, TheraMab Bioscience [2]. Some newcomers later joined the sector, such as Rongcan Biotech, RiboBio, YolTech Therapeutics, CirCode Bio, Everest Medicines, CSPC Pharmaceutical Group, and Sinopharm Group.
Completely contrary to public perceptions, Communist China had been preparing for mRNA technology research for years. During an interview, the founder of RNACure Biopharma Yu Hang suggested he witnessed the growth of Moderna and planned to enter the mRNA field in 2016 after being inspired. [3] Founded by Dr. Li Hangwen in 2016, Stemirna Therapeutics was the first company to bring mRNA technology into China, and this company began development of MERS (Middle East Respiratory Syndrome) and influenza mRNA vaccines in 2017. [4] Fudan University Professor Lin Jinzhong, who co-founded the Shanghai mRNA Innovation and Translation Center, began research on mRNA technology in 2017. [5] CanSino Bio started mRNA research in 2018 and developed a software platform to optimize mRNA sequences. [6]

Screenshot 1 of Stemirna Therapeutics’ company website. [4] The webpage was deleted.

Screenshot 2 of Stemirna Therapeutics’ company website. [4]
Communist China conducted human clinical trials of mRNA therapeutics since long ago. A timeline on the website of Stemirna Therapeutics indicated it carried out clinical trials of an anti-cancer mRNA vaccine on cancer patients in 2018. (The company’s website was suddenly deleted a few days ago). [4] If it started clinical trials of mRNA therapeutics at such an early time, did it discover any adverse effects?
Fudan University is the first university in China to research on mRNA technology. [5] Several bellwethers in this field are from Fudan University. For example, founder of Fosun Pharma Guo Guangchang, CEO of Junshi Pharma Li Ning, and Director of the National Infectious Disease Medical Center Zhang Wenhong (nicknamed China’s Fauci) are all Fudan University alumni. Lin Jinzhong is a professor at Fudan University. [7] Zhang Wenhong often advocates for Covid jabs, but he is also listed as an expert at the Shanghai mRNA Innovation and Translation Center [5]—he is also a link on the vaccine production chain.
Some Chinese pharmaceutical companies relied on foreign technology to develop mRNA Covid vaccines. The most well-known example is the agreement between BioNTech and Fosun Pharma. In addition, in May 2020, CanSino Bio bought the technology of Canadian company Precision NanoSystems to develop an mRNA vaccine. [6] In September 2021, Everest Medicines licensed in mRNA technology from another Canadian company Providence Therapeutics. [8] Besides relying on foreign technology, Communist China also possesses robust research capability on mRNA technology. There are many mRNA tech companies in Communist China, and its research capacity is as strong as Western big pharma.
To some sense, Communist China’s mRNA technology is obtained from the United States or Western countries. Many top scientists have work experiences in Western research institutions or companies. To name a few, founder of Stemirna Therapeutics Li Hangwen earned his PhD degree at the MD Andersen Cancer Center [4, 9]. Fudan University professor Lin Jinzhong was a postdoc at Yale University [10]. Founder of InnoRNA Li Linxian’s postdoc mentor at MIT was the founder of Moderna [9]. Founder of ImmoRNA Biotechnology Wang Zihao used to work at GSK [11]. For Rongcan Biotech founder Zhang Xueqing, her mentor was also the founder of Moderna [12, 13]. Founder of TheraMeb Bioscience Wu Chenyan used to be a senior scientist at Pfizer [14]. Founder of Abogen Biosciences Ying Bo was a former employee of Moderna [15]. These scientists made further improvements upon Western technology and claimed to own intellectual property. (Wu Chenyan was indicted in the United States for stealing Pfizer’s mRNA data, though [16].)
Communist China’s mRNA startups were established at a strange time point. Six of the nine early mRNA vaccine startups were founded in 2019, in the months leading up to the Covid pandemic. (They are Abogen Biosciences, LiveRNA, RNACure Biopharma, InnoRNA, ImmoRNA Biotechnology, and Rhegen Biomedical Technology.) [2] In most of 2019 before the pandemic, mRNA technology was an obscure and unprofitable field since vaccine demand was low. So many mRNA vaccine companies popped up together in Communist China in 2019, and this pattern probably indicates some kind of pre-planning. Then the pandemic erupted afterward.
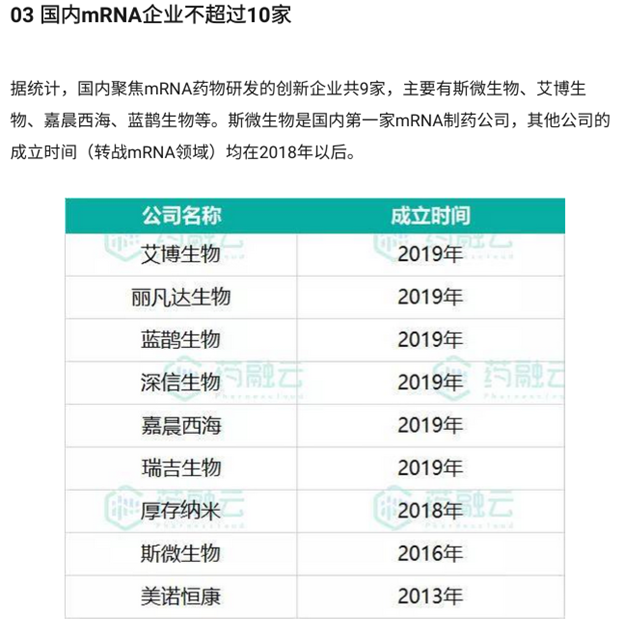
Founding time of early mRNA startups. Image source: [2]
Some tech startups were tied to Communist China’s military. Abogen Biosciences partnered with Walvax Biotechnology and the Academy of Military Medical Sciences (AMMS) to create ARCoV vaccine, which is Communist China’s first domestic mRNA Covid vaccine to enter clinical trials [14]. LiveRNA collaborated with AMMS in mRNA vaccine development [14]. TheraMeb Bioscience carried out animal studies on mRNA Covid vaccine at the AMMS [14]. CanSino Bio also worked with Communist China’s military.
Some startups were supported by giant capital funds. Abogen Biosciences had undergone several rounds of fundraising from investors, one of which was Boyu Investment [17]. At early phase, the sole sponsor of Rhegen Biomedical Technology was Sequoia China [18].
Besides the Fosun Pharma’s licensed BioNTech vaccine, six brands of mRNA Covid vaccines have entered clinical trials. The six brands are Abogen-Walvax-AMMS partnership, Stemirna Therapeutics, LiveRNA, RiboBio-Agena Bioscience partnership, CSPC Pharmaceutical Group, and CanSino Bio. [19] The CSPC vaccine is already granted EUA.
Believe or not, Communist China developed mRNA Covid vaccines at lightning speed. The country has developed several types of mRNA vaccine within a few months of the Covid outbreak, although the progress of clinical trials and public usage was slow. In spring 2020, founder of LiveRNA Peng Yucai designed an mRNA vaccine candidate within eight days, and the design was submitted to government agencies for animal tests on February 1, 2020. [14] Abogen’s ARCoV vaccine was approved for phase I clinical trials in June 2020. [20] Lin Jinzhong and RNACure Biopharma used mRNA vaccine to produce virus-like particles and submitted their research results to the journal Cell Research in June 2020 [21], but Lin already attained experimental success in February 2020 [3].
A key feature of Communist China’s domestic mRNA vaccines is stability under high temperature. Both Abogen’s ARCoV vaccine and CSPC Group’s SYS6006 vaccine could be stored at 2-8 degrees Celsius for months, obviating the need for freezer storage [22, 23]. Rhegen Biomedical Technology made further progress by the invention of a lyophilized mRNA vaccine dry powder that is stable at room temperature (25 degrees Celsius) [24, 25]. In my opinion, such vaccines have pros and cons. The upside is that chemicals in the vaccines are stable and resistant to degradation, and are therefore convenient for transportation and storage. But the downside is whether the chemicals are bio-degradable after injection into the human body. Will the chemical residues cause adverse effects if they cannot be removed in the human body?
Even some Chinese media questioned the safety of domestically produced mRNA vaccines. The phase I clinical trials report of Abogen’s ARCoV concluded the vaccine was safe and effective. However, careful scrutiny of the experimental data revealed points of concern. About 90% of the 15-microgram dosage group reported adverse effects, of which 85% were fevers. The 25-microgram dosage group had 100% fever rate [26, 27]. This data proves that mRNA vaccines could be harmful for humans. Moreover, during an interview, founder of Longuide Zhang Longgui explained that lipid nanoparticles (LNP) exhibit dosage-dependent toxicity and are therefore unsuitable for high-dose and long-time injections [28]. Anyway, mRNA vaccines are somewhat toxic.
The pharmaceutical executives sometimes attended scientific seminars together. On March 11, 2021, the Shanghai Biopharmaceutics Industry Association hosted the mRNA Pharmaceutical Innovation Seminar. To name a few, guests of this event were Fosun Pharma’s President of Global R&D Hui Aimin, Professor Lin Jinzhong of Fudan University, founder of Stemirna Therapeutics Li Hangwen, and founder of RNACure Biopharma Yu Hang. [29]
These mRNA companies excel in different technological strong points. Some are even unicorns with unique technology, at least in China. For instance, (1) Stemirna’s mRNA vaccines use Lipopolyplex (LPP) instead of lipid nanoparticles (LNP) for delivery. In an LPP, polymers first encapsulate the mRNA to form a core that is in turn coated by lipid, so that there are two layers of coatings. [19] (2) ImmoRNA Biotechnology is the only company in China that develops self-replicating mRNA, which could self-replicate inside cells like viruses, as the name suggests. Self-replicating mRNA is a very potent drug. [19] (3) Rhegen Biomedical Technology pioneered lyophilized mRNA vaccine dry powder that is stable at room temperature (25 degrees Celsius) [19] (4) YolTech Therapeutics specializes in combining gene-editing CRISPR-Cas9 technology with mRNA technology, employing lipid nanoparticles to deliver gene-editing mRNA. [19] (5) CirCode Bio develops circular RNA that is resistant to degradation. [30]
No matter how advanced the technology is, mRNA vaccines are NOT free of adverse effects. Don’t take the mRNA jabs if you want a long and healthy life.
References List (Sorry, most of the references are webpages written in Chinese): Click Here to Copy and Paste
不是沃森生物! 石药集团拿下首款国产mRNA新冠疫苗, 从临床到获批11个月。凤凰新闻。2023-03-23
https://finance.ifeng.com/c/8OOJXl5fV2a
2. 国产mRNA疫苗现状, 嘉晨西海成”海王”, 国内mRNA企业不超10家。搜狐网。2021-07-26
https://www.sohu.com/a/479594694_121035734
经纬人物No.21-蓝鹊生物创始人俞航。雪球网。2021-08-27
https://xueqiu.com/7211544918/195815828
斯微生物官网 (网页突然被删除)
https://www.stemirna.com/History/list.aspx
张江mRNA国际创新中心官网
https://www.shmitc.org.cn/aboutus/superiority
详解康希诺和石药的新冠mRNA疫苗及其辅料国产化。雪球网。2022-04-28
https://xueqiu.com/1036022495/218700743
复星集团到底干什么的? 这么有钱? 知乎网。2022-09-24
https://www.zhihu.com/question/280582791/answer/2641787247?utm_id=0
履新云顶新耀后, 罗永庆首谈心路历程和公司规划。腾讯新闻。2023-01-06
https://view.inews.qq.com/k/20230106A09Y7Z00?no-redirect=1&web_channel=wap&openApp=false
时代的箭头: mRNA疫苗背后的中国力量。中国医药创新促进会。2021-06-08
http://www.phirda.com/artilce_24549.html?cId=1
林金钟教授简介。复旦大学官网。
https://life.fudan.edu.cn/b3/74/c28175a308084/page.htm
嘉晨西海官网
https://www.immorna.com/cn/about.html
LNP专利, mRNA制药产业的痛点。雪球网。2021-12-16
https://xueqiu.com/4376152234/206091927
章雪晴教授简介。上海交大官网。
https://pharm.sjtu.edu.cn/szdy/2639.html
mRNA三大巨头撑起约6000亿市值, 国内mRNA公司一览。雪球网。2020-12-27
https://xueqiu.com/5603603160/166878330
15. 艾博生物创始人英博: 不只新冠疫苗, 立足mRNA技术平台的产业变革。雪球网。2021-09-06
https://xueqiu.com/1916655314/196851670
一家mRNA创新药企的猝死之谜。网易。2022-05-27
https://m.163.com/dy/article/H8DCUUL90514BJBO.html?spss=adap_pc
艾博生物官网
https://www.abogenbio.com/news2021/140.html
瑞吉生物, 敢做创业路上的孤勇者-红杉早期医疗投资特写。搜狐网。2022-09-09
https://www.sohu.com/a/583568130_121119001
全面梳理国内mRNA研发管线。健康界。2022-04-13
https://www.cn-healthcare.com/articlewm/20220414/wap-content-1338297.html
Zhang NN, and et al. A Thermostable mRNA Vaccine against COVID-19. Cell. 2020 Sep 3;182(5):1271-1283.e16.
doi: 10.1016/j.cell.2020.07.024. Epub 2020 Jul 23. PMID: 32795413; PMCID: PMC7377714.
https://pubmed.ncbi.nlm.nih.gov/32795413/
Lu J, and et al. A COVID-19 mRNA vaccine encoding SARS-CoV-2 virus-like particles induces a strong antiviral-like immune response in mice. Cell Res. 2020 Oct;30(10):936-939.
doi: 10.1038/s41422-020-00392-7. Epub 2020 Aug 17. PMID: 32801356; PMCID: PMC7429369.
https://pubmed.ncbi.nlm.nih.gov/32801356/
Zhao H, and et al. Long-term stability and protection efficacy of the RBD-targeting COVID-19 mRNA vaccine in nonhuman primates. Signal Transduct Target Ther. 2021 Dec 24;6(1):438.
doi: 10.1038/s41392-021-00861-4. PMID: 34952914; PMCID: PMC8703211.
https://pubmed.ncbi.nlm.nih.gov/34952914/
康希诺, 石药集团mRNA疫苗分别获批临床, 盘点国内mRNA疫苗研发公司。健康界。2022-04-06
https://www.cn-healthcare.com/articlewm/20220405/wap-content-1334712.html
Ai L, and et al. Lyophilized mRNA-lipid nanoparticle vaccines with long-term stability and high antigenicity against SARS-CoV-2. Cell Discov. 2023 Jan 23;9(1):9. doi: 10.1038/s41421-022-00517-9. PMID: 36683074; PMCID: PMC9868121.
https://pubmed.ncbi.nlm.nih.gov/36683074/
全球首款冻干mRNA疫苗, 来自中国团队, 可在25度下长期稳定。健康界。2022-02-19
https://www.cn-healthcare.com/articlewm/20220215/wap-content-1314995.html
艾博生物新冠疫苗合作方沃森生物”牵手”新伙伴, 公司回应为完善产品布局。每日经济新闻。2022-01-29
https://m.nbd.com.cn/articles/2022-01-29/2111710.html
Chen GL, and et al. Safety and immunogenicity of the SARS-CoV-2 ARCoV mRNA vaccine in Chinese adults: a randomised, double-blind, placebo-controlled, phase 1 trial. Lancet Microbe. 2022 Mar;3(3):e193-e202. doi: 10.1016/S2666-5247(21)00280-9. Epub 2022 Jan 24. PMID: 35098177; PMCID: PMC8786321.
https://pubmed.ncbi.nlm.nih.gov/35098177/
癌细胞可一针清除? mRNA引爆又一场疫苗革命, 四年前无投资者看好。新浪财经。2022-10-28
https://finance.sina.cn/2022-10-28/detail-imqmmthc2447288.d.html?from=wap
mRNA专家齐聚! 快来看mRNA药物创新论坛嘉宾阵容和日程。药时代。2021-03-09
https://www.drugtimes.cn/2021/03/09/6aa0d5a6bc/
环码生物官网
http://m.circodebio.cn/
Source: Gnews,Twitter, UPI(Image)
Also Read:
