A recent disclosure by a Swedish journalist on the social media platform X has triggered significant public discourse regarding the safety of COVID-19 vaccines. The journalist reported that a senior official from Pfizer admitted during a testimony to an inquiry commission that approximately 60,000 deaths may be attributable to the vaccine’s side effects. This shocking claim has yet to gain traction in mainstream media, which the journalist accuses of being tightly controlled by influential entities, including networks linked to Bill Gates and the pharmaceutical industry, thereby suppressing crucial information from the public.
Media Silence and Public Reaction
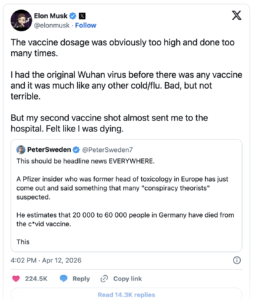
Despite the notable lack of coverage from traditional news outlets, the journalist’s tweet rapidly gained attention, amassing over 25 million views within six hours. This reflects a significant public interest and a burgeoning concern about vaccine safety. The discussion intensified further when Elon Musk shared the post, recounting his own negative experience following vaccination. Musk’s amplification contributed an additional 24 million views, escalating the already heated conversation into a more substantial global debate.
This situation extends beyond a simple public health issue; it raises critical questions about the control of information, narrative manipulation, and the systematic silencing of dissenting views. Such admissions, which have been overlooked by major publishers, call into question the broader implications of how information and public health discourse are managed. The experience of sudden cardiac events in vaccinated younger populations highlights the need for urgent and thorough investigations into the potential side effects of these vaccines.
The alarming patterns of adverse health events linked to COVID-19 vaccinations underscore the urgent need for transparent, independent investigation. The absence of an open dialogue on these pressing concerns has sparked calls for accountability. If pharmaceutical interests and their affiliated networks are influencing the information presented to the public, the implications extend into the realm of moral responsibility. It is suggested that every life lost and every question raised demands thorough scrutiny and action.
The messaging is clear: protecting human life should surpass the interests of profit or reputation. The ramifications of suppressing such critical information can no longer be dismissed as mere ignorance; they represent a profound complicity in the system that prioritizes convenience over truth.
Inquiry Background
Pfizer Toxicologist’s Bombshell Testimony Rocks German Parliament

In March 2026, Germany’s Bundestag hosted a parliamentary inquiry into the safety and efficacy of COVID-19 vaccines, prompted by ongoing public concerns over adverse events and data transparency. Dr. Helmut Sterz, a veteran toxicologist who served as Pfizer’s chief toxicologist for Europe until 2005, emerged as a key witness. His 30+ years at the company gave him insider knowledge of pharmaceutical testing protocols, making his statements particularly weighty.

Dr. Helmut testified that Pfizer’s mRNA vaccine (Comirnaty) bypassed standard toxicology testing required for novel products. He highlighted:
- Lack of Preclinical Tox Studies: No comprehensive animal trials assessed lipid nanoparticle distribution or long-term toxicity before human rollout. “This was unprecedented,” Sterz stated, comparing it to routine drug development where such tests are mandatory.
- Batch Variability: Internal data showed huge differences in adverse event rates across vaccine batches—some “hot lots” linked to severe reactions, while others were benign. He called this “a red flag ignored.”
- Underreported Risks: Pfizer allegedly prioritized speed over safety, with mechanisms like the PREKINE study (a small Phase I trial) insufficient to catch organ accumulation of spike proteins or lipids.
These claims align with whistleblower documents and VAERS data analyses, which Dr. Helmut referenced, noting ratios of severe events up to 12:1 between high- and low-risk batches.

Dr. Helmut accused Pfizer of “scientifically untenable” shortcuts, urging independent batch testing. He criticized the EMA and Paul Ehrlich Institute for rubber-stamping approvals without demanding full tox data. “Patients were guinea pigs,” he said bluntly.
- Carcinogenicity and Reproductive Toxicity Not Tested:
- The carcinogenic risk of Comirnaty was not evaluated prior to approval due to time constraints.
- No alternative studies were conducted to compensate for this omission.
- A reproductive toxicity study on rats was poorly conducted, yielding unreliable data on effects during pregnancy and fetal development.
- Accelerated Approval Process:
- Approval was granted rapidly (“Schnelldurchgang”) according to the Robert Koch Institute (RKI) internal protocols.
- Essential toxicological studies were sacrificed without valid justification.
- Dr. Helmut states no comparable vaccine with a similar indication has been approved without these studies.
- This expedited approval led to what the expert terms “illegal human experiments”.
- Post-Marketing Safety Surveillance and Mortality Data:
- Pfizer’s postmarketing report indicated over 1,200 suspected deaths within two months after approval.
- The Paul Ehrlich Institute (PEI) received 2,133 death reports post-Comirnaty vaccination in Germany, but underreporting is significant.
- In the U.S., an underreporting factor of about 30 is assumed; applying this to Germany suggests up to 60,000 vaccine-related deaths, though this figure is extrapolated and thus Not specified/Uncertain.
- The German government withheld key safety and vaccination data from commissions, many of whom declined to request it.
- Legal and Risk-Benefit Considerations:
- Many vaccine-injured patients in Germany struggle to obtain compensation; courts often rule that Comirnaty has a positive benefit-risk ratio.
- Clinical trials did not assess Comirnaty’s ability to prevent severe COVID-19 or death directly.
- A mathematical estimate suggests that for every severe COVID case prevented, about 25 severe adverse reactions occur.
- Mortality Trends:
- Age-adjusted mortality in Germany increased in 2021 and 2022 compared to 2020, contradicting expectations if the vaccine were effective and safe.
- Vaccine Composition and Manufacturing Concerns:
- The vaccine tested in clinical trials was a highly purified substance too costly for mass production.
- The widely distributed vaccine was produced using the bacterium Escherichia coli, resulting in significant contamination with bacterial DNA.
- This contamination may contribute to a substantially increased cancer risk.
The testimony, livestreamed and widely shared online, has fueled calls for vaccine recalls, lawsuits, and a EU-wide audit. Opposition MPs hailed it as “explosive”.
This fits a pattern of 2026 revelations, including U.S. lawsuits against Pfizer for misrepresenting efficacy (95% claim based on relative risk reduction) and Finnish data showing excess mortality post-vaccination.
The revelations surrounding the possible side effects of the COVID-19 vaccine, alongside the media’s apparent reluctance to cover these stories, urge a reevaluation of the dynamics at play in public health decision-making. The public’s demand for truth and answers is rising, indicating a crucial moment in which ordinary citizens are seeking more transparent discourse regarding vaccine safety. It is imperative that stakeholders—governments, pharmaceutical companies, and media organizations—be held accountable for the information shared, as collective human life and wellbeing are at stake.
Also read:
Genetic Apocalypse: Pfizer’s mRNA Deception and the Threat to Humanity
Pfizer Caught Funnelling $12 Million to Anderson Cooper To Promote mRNA Jabs to Americans
Dr. Naomi Wolf Uncovers Pfizer’s Depopulation Agenda, as Evidenced by Its Own Documents
